The benefit of stainless steel furniture is that it’s tough and long-lasting. In addition, our 304 and 316 ranges have excellent corrosion resistance due to the formation of a chromium passivation layer. This makes them suitable for use in hygiene-conscious environments such as the food processing and pharmaceutical sectors.
It's important to understand that stainless steel must contain at least 10.5% chromium to form a passive layer on the surface: the more chromium content in the stainless steel, the better its corrosion resistance. In comparison, our 304 stainless steel contains 18-20% chromium.
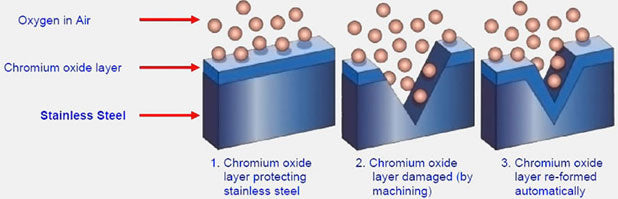
This chromium forms an invisible passive layer of chromium oxide on the stainless steel surface to protect itself from exposure to contaminants and therefore corrosion. If the surface is physically (due to scratches or dents) or chemically damaged, the ion in the stainless steel is exposed which causes the staining (rouging). The passive layer will repair itself once the source of the damage is removed and chemical treatment has been used. A very clean surface is a critical prerequisite to the formation of this passive layer and chemical treatments can enhance the passive layer.
We've created a handy stainless steel care guide to help advise on your cleaning needs.